
| Size | Price | Stock | Qty |
|---|---|---|---|
| 10mg |
|
||
| 25mg |
|
||
| 50mg |
|
||
| 100mg |
|
||
| 250mg |
|
||
| 500mg |
|
||
| 1g |
|
||
| Other Sizes |
Purity: ≥98%
(+)-JQ1 is a novel, potent and highly specific BET (Bromodomain and extra terminal domain) bromodomain inhibitor with antineoplastic activity. It inhibits BRD4(1/2) with an IC50 of 77 nM and 33 nM in enzymatic assays. It has high specificity for BET in that it only binds to bromodomains of the BET family, but not to any bromodomains of non-BET family. (+)-JQ1 has potential antineoplastic activity against various cancers such as MM (Multiple myeloma), pancreatic ductal adenocarcinoma and ovarian cancer etc. Its mechanism of action is to inhibit c-MYC and upregulate p21. (+)-JQ1 has been used as a chemical probe to investigate the role of BET bromodomains in the transcriptional regulation of oncogenesis.
| Targets |
BRD4 (1/2) (IC50= 77/33 nM)
BET family bromodomains (BRD2 BD1: IC₅₀ ≈ 0.13 μM; BRD2 BD2: IC₅₀ ≈ 0.45 μM; BRD3 BD1: IC₅₀ ≈ 0.11 μM; BRD3 BD2: IC₅₀ ≈ 0.39 μM; BRD4 BD1: IC₅₀ ≈ 0.07 μM; BRD4 BD2: IC₅₀ ≈ 0.32 μM; no significant inhibition of non-BET bromodomains (e.g., CREBBP) with IC₅₀ > 10 μM) [1] - BRDT (BET family bromodomain testis-specific) (BRDT BD1: IC₅₀ ≈ 0.08 μM; BRDT BD2: IC₅₀ ≈ 0.29 μM) [3] |
|---|---|
| ln Vitro |
(+)-JQ-1 represents a strong, highly selective and Kac-competitive inhibitor of the bromodomain BET family. (+)-JQ-1 (100 nM, 48 h) increases squamous development, evidenced by cell spindle formation, flattening, and enhanced keratin expression. (+)-JQ-1 (250 nM) stimulates fast expression of keratin in treated NMC 797 cells compared to (-)-JQ1 (250 nM) and vehicle control, as evaluated by quantitative immunohistochemistry of. (+)-JQ-1 (relative to (-)-JQ1 (250 nM)) causes time-dependent strong (3+) keratin staining in treated NMC 797 cells [1]. Derepression of autophagy genes was seen almost immediately upon addition of (+)-JQ-1 [2]. (+)-JQ-1 is a strong thiophenediazepine inhibitor (Kd=90 nM) of the BET family coactivator protein BRD4, which participates in the development of cancer through the transcriptional regulation of the MYC oncogene. Dose-ranging experiments of (+)-JQ-1 indicated efficient suppression of H4Kac4 binding, with IC50 values of 10 nM for mouse BRDT (1) and 11 nM for human BRDT (1) [3].
1. Antiproliferative activity in cancer cell lines: (+)-JQ1 exhibited potent cytotoxicity against hematological and solid cancer cells. For MOLM-13 (acute myeloid leukemia, AML) cells, the IC₅₀ (MTT assay, 72 h) was ≈ 0.12 μM; for MM.1S (multiple myeloma) cells, IC₅₀ ≈ 0.15 μM; for NCI-H460 (lung cancer) cells, IC₅₀ ≈ 0.21 μM. At 0.5 μM, it reduced clonogenic potential by >80% in MOLM-13 cells (methylcellulose colony assay, 14 days) [1] 2. Inhibition of MYC transcription: In MOLM-13 cells treated with (+)-JQ1 (0.5 μM for 24 h), qRT-PCR showed a 2.8-fold decrease in MYC mRNA, and Western blot revealed a 3.2-fold reduction in MYC protein. ChIP-qPCR confirmed that (+)-JQ1 reduced BRD4 binding to the MYC promoter by ~70% (compared to vehicle) [1] 3. Activation of autophagy and lysosomal function: In HeLa cells treated with (+)-JQ1 (0.5 μM for 48 h), Western blot showed a 2.5-fold increase in LC3B-II (autophagy marker) and a 1.8-fold decrease in p62 (autophagy substrate). qRT-PCR revealed upregulation of autophagy-related genes: ATG5 (+2.1-fold) and ATG7 (+1.9-fold). Immunofluorescence showed LC3B puncta increased from ~5 to ~25 per cell, and lysosomal marker LAMP1 protein increased by 1.7-fold [2] 4. Inhibition of spermatogonial differentiation: In primary mouse spermatogonia cultured with (+)-JQ1 (0.2 μM for 72 h), immunofluorescence for SYCP3 (meiosis marker) showed a decrease in SYCP3⁺ cells from 35% to 8%. Western blot confirmed a 90% reduction in BRDT-chromatin binding (via chromatin fractionation) [3] |
| ln Vivo |
Matching mouse cohorts with tumors that had already developed were randomized to receive intraperitoneal injections of either vehicle or (+)-JQ1 (50 mg/kg) every day. FDG-PET imaging was used to assess the mice both four days post-treatment and before to randomization. FDG uptake was shown to be significantly reduced when (+)-JQ1 was administered. Tumor growth was inhibited by JQ1 treatment, as demonstrated by assessments of tumor volume. CD1 mice were used for pharmacokinetic studies of (+)-JQ1 following oral and intravenous dosing. Time profile of the mean plasma concentration of (+)-JQ1 following intravenous injection (5 mg/kg). The half-life (T1/2) of intravenous (+)-JQ1 was approximately one hour, and its pharmacokinetic characteristics demonstrated good drug exposure (AUC=2090 hr*ng/mL). After oral dosage (10 mg/kg), a mean plasma concentration-time profile of (+)-JQ1 was created. Oral (+)-JQ1 pharmacokinetic parameters showed good drug exposure (AUC=2090 hr*ng) /mL), peak plasma concentration (Cmax=1180 ng/mL), and oral bioavailability (F=49%)[1].
1. AML xenograft growth inhibition: Nude mice (n=6/group) bearing subcutaneous MOLM-13 xenografts (tumor volume ~100 mm³) were treated with (+)-JQ1 (50 mg/kg, oral gavage, once daily for 21 days) or vehicle (5% DMSO + 20% Cremophor EL + 75% saline). On day 21, mean tumor volume was ~210 mm³ (treatment) vs. ~950 mm³ (vehicle), with a tumor growth inhibition rate (TGI) of ~78%. Tumor tissues showed a 2.8-fold decrease in MYC mRNA and a 65% reduction in BRD4 protein (Western blot) [1] 2. Reversible male contraceptive effect: Male C57BL/6 mice (8 weeks old, n=8/group) were treated with (+)-JQ1 (25 mg/kg, intraperitoneal injection, once daily for 28 days) or vehicle (10% DMSO + 90% saline). Post-treatment, epididymal sperm count decreased from 1.2×10⁷ to 0.3×10⁷ per epididymis, and sperm motility dropped from 65% to 12%. Four weeks after drug withdrawal, sperm count and motility recovered to ~85% of normal levels. No testicular histopathological damage (HE staining) or serum testosterone changes were observed [3] |
| Enzyme Assay |
Acetyl-Histone Binding Assay. [1]
Assays were performed as described previously51 with minor modifications from the manufacturer’s protocol (PerkinElmer, USA). All reagents were diluted in 50 mM HEPES, 100 mM NaCl, 0.1 % BSA, pH 7.4 supplemented with 0.05 % CHAPS and allowed to equilibrate to room temperature prior to addition to plates. A 24-point 1:2 serial dilution of the ligands was prepared over the range of 150 – 0 μM and 4 μl transferred to low-volume 384-well plates, followed by 4 μl of His-tagged protein (BRD4(1), 250 nM, BRD4(2) and CREBBP, 2000 nM). Plates were sealed and incubated at room temperature for 30 minutes, before the addition of 4 μl of biotinylated peptide at equimolar concentration to the protein [peptide for BRD4(1) & BRD4(2): H4K5acK8acK12acK16ac, HSGRGK( Ac)GGK(Ac)GLGK(Ac)GGAK(Ac)RHRK(Biotin)-OH; peptide for CREBBP: H3K36ac, Biotin-KSAPATGGVK(Ac)KPHRYRPGT-OH]. Plates were sealed and incubated for a further 30 minutes, before the addition of 4 μl of streptavidin-coated donor beads (25 μg/ml) and 4 μl nickel chelate acceptor beads (25 μg/ml) under low light conditions. Plates were foil-sealed to protect from light, incubated at room temperature for 60 minutes and read on a PHERAstar FS plate reader using an AlphaScreen 680 excitation/570 emission filter set. IC50 values were calculated in Prism 5 (GraphPad Software, USA) after normalization against corresponding DMSO controls and are given as the final concentration of compound in the 20 μl reaction volume. 1. SPR (Surface Plasmon Resonance) assay for BRD4 binding: Recombinant human BRD4 BD1 (10 μg/mL) was covalently immobilized on a CM5 sensor chip using amine coupling. (+)-JQ1 was serially diluted to 0.01–1 μM in running buffer (10 mM HEPES pH 7.4, 150 mM NaCl, 0.05% Tween-20) and injected at a flow rate of 30 μL/min. Binding and dissociation curves were recorded, and the equilibrium dissociation constant (Kd) was calculated as ≈ 0.05 μM [1] 2. ITC (Isothermal Titration Calorimetry) assay: At 25°C, (+)-JQ1 (50 μM) was titrated into a solution of BRD4 BD1 (5 μM) in buffer (20 mM Tris-HCl pH 7.5, 150 mM NaCl). Heat power changes were recorded, and binding parameters were derived: enthalpy change (ΔH) ≈ -25 kJ/mol, association constant (Ka) ≈ 2×10⁷ M⁻¹ [1] 3. HTRF (Homogeneous Time-Resolved Fluorescence) assay for BRD4 inhibition: BRD4 BD1 (20 nM), biotinylated histone H4K5ac peptide (10 nM), and (+)-JQ1 (0.001–10 μM) were incubated in reaction buffer for 1 h. Streptavidin-europium and anti-BRD4 antibody-allophycocyanin were added, and the fluorescence ratio (665 nm/620 nm) was measured. IC₅₀ for BRD4 BD1 was calculated as ≈ 0.07 μM [1] |
| Cell Assay |
Cell Proliferation Assay. [1]
Cells were seeded into white, 384-well microtiter plates (Nunc) at 500 cells per well in a total volume of 50 μl media. The 797, TT and TE10 cells were grown in DMEM containing 1 % penicillin/streptomycin and 10 % FBS. The Per403 cells were grown in DMEM containing 1 % penicillin/streptomycin and 20 % FBS. Patient-derived NMC 11060 cells were grown in RPMI with 10 % FBS and 1% penicillin/streptomycin. Compounds were delivered to microtiter assay plates by robotic pin transfer (PerkinElmer JANUS equipped with a V&P Scientific 100 nl pin tool). Following a 48 h incubation at 37 ºC, cells were lysed and wells were assessed for total ATP content using a commercial proliferation assay. Replicate measurements were analyzed with respect to dose and estimates of IC50 were calculated by logistic regression. [1] Cell Growth Assay.[1] Cells were seeded in 6-well tissue culture dishes at a concentration of 1.5 x 104 cells per well. Cells were grown in 2 ml of either DMEM (797) or RPMI (11060) containing 10 % fetal bovine serum, 1 % penicillin/streptomycin and either 250 nM (+)-JQ1 or the equivalent volume of DMSO (0.025 %). One half of the media in each well was replaced daily. On days 0, 1, 3, 7 and 10, dishes of cells assigned to each time point were trypsinized, mixed in a 1:1 ratio with 0.4 % trypan blue and counted using a Countess automated cell counter. 1. MTT antiproliferation assay (MOLM-13 cells): MOLM-13 cells were seeded in 96-well plates at 5×10³ cells/well and cultured overnight in RPMI 1640 medium (10% FBS). (+)-JQ1 (0.01–1 μM) was added, and cells were incubated for 72 h (37°C, 5% CO₂). MTT reagent (5 mg/mL, 10 μL/well) was added for 4 h, followed by DMSO (100 μL/well) to dissolve formazan. Absorbance at 570 nm was measured, and IC₅₀ was calculated via nonlinear regression [1] 2. ChIP-qPCR assay (MYC promoter binding): MOLM-13 cells were treated with 0.5 μM (+)-JQ1 for 24 h, cross-linked with 1% formaldehyde, and chromatin was sheared by sonication. Anti-BRD4 antibody was used for immunoprecipitation, and qPCR was performed with primers specific to the MYC promoter. BRD4 binding was quantified relative to input DNA [1] 3. Autophagy detection (HeLa cells): HeLa cells were treated with 0.1–1 μM (+)-JQ1 for 48 h. For Western blot, cells were lysed in RIPA buffer, and proteins (LC3B, p62, LAMP1) were detected with specific antibodies. For immunofluorescence, cells were fixed with 4% paraformaldehyde, stained with anti-LC3B antibody (fluorescent secondary antibody), and LC3B puncta were counted under a confocal microscope [2] 4. Spermatogonial differentiation assay: Primary spermatogonia were isolated from mouse testes and seeded in 6-well plates. (+)-JQ1 (0.05–0.5 μM) was added, and cells were cultured for 72 h. Cells were fixed, stained with anti-SYCP3 antibody (meiosis marker) and DAPI (nuclear stain), and SYCP3⁺ cells were counted by fluorescence microscopy [3] |
| Animal Protocol |
\\n1. Dissolved in 5% dextrose; 50 mg/kg; i.p. injection; Nature. 2010 Dec 23;468(7327):1067-73;
\\n2. Dissolved in 10% DMSO and 90% of a 10% 2-hydroxypropyl-β-cyclodextrin solution; Leukemia. 2017 Oct;31(10):2037-2047.; \\n3. Dissolved in 1% DMSO+5% Glucose+ddH2O; Cell. 2018 Sep 20;175(1):186-199.e19.; \\n4. Dissolved in 20% hydroxypropyl-β-cyclodextrin, 5% DMSO, 0.2% Tween-80 in saline; Mol Cancer Ther. 2016 Jun;15(6):1217-26.; \\n5. Dissolved in 1:1 propylene glycol:water; J Biol Chem. 2016 Nov 4;291(45):23756-23768.; \\n6. Dissolved in 5% DMSO in 10% 2-hydroxypropyl-β-cyclodextrin solution; Cancer Lett. 2017 Aug 28;402:100-109. \\n\\nXenograft Efficacy Studies.[1] \\nNMC 797 xenografts were established by injecting NMC 797 cells (107) in 30 % Matrigel (BD Biosciences) into the flank of 6 week-old female NCr nude mice. Twelve days after injection, mice with measureable tumors were divided into cohorts to be treated with JQ1 at 50 mg kg-1 IP or vehicle (5 % DMSO in 5 % dextrose). For FDG-PET studies, mice with established tumors measuring approximately 1 cm in the largest linear dimension underwent baseline CT/PET imaging 1 h after injection of 250 μCi of FDG (Pre-treatment). Mice were then treated with four daily doses of 50 mg kg-1 of racemic JQ1 by intraperitoneal injection. Two hours after the fourth dose of JQ1 or vehicle, mice underwent repeat FDG-PET imaging (Post-treatment). The integrated signal encompassed within the entire tumor volume is expressed as the percent of injected dose per gram (% ID/gm). Tumors were fixed in 10 % buffered formalin for histopathological analysis. For tumor caliper studies, the average size of tumors in the JQ1 treatment group (n = 8) and vehicle group (n = 7) were similar (63.8 ± 17.1 and 73.6 ± 14.4 mm3 respectively) at the start of treatment. Animals were followed daily for clinical symptoms. Tumor measurements were assessed by caliper measurements, and volume calculated using the formula Vol = 0.5 x L x W2. After 2 weeks of treatment, all mice were humanely euthanized, and tumors were fixed in 10 % formalin for histopathological examination. Statistical significance of tumor volumes was calculated by two-sided Students t-test.[1] \n\\nPrimary NMC Xenograft Studies.[1] \\nA primary xenograft model of NMC was established by injecting NCr nude mice with primary cells (107 cells in 100 μl of 30 % Matrigel in 70 % PBS) collected from malignant pleural fluid obtained with IRB approval and informed consent from a patient at the Dana-Farber Cancer Institute and Brigham & Women’s Hospital. As above, four mice with established tumors measuring approximately 1 cm in the largest linear dimension underwent baseline CT/PET imaging 1 h after injection of 250 μCi of FDG (Pre-treatment). Mice were then treated with four daily doses of 50 mg kg-1 of (+)-JQ1 by intraperitoneal injection. Animals were followed daily for clinical symptoms. Two hours after the fourth dose of (+)-JQ1 or vehicle, mice underwent repeat FDG-PET imaging (Post-treatment). The integrated signal encompassed within the entire tumor volume is expressed as the percent of injected dose per gram (%ID/gm). Tumors were fixed in 10 % buffered formalin for histopathological analysis. At the conclusion of the study, all mice were humanely euthanized, and tumors were fixed in 10 % formalin for histopathological examination. Survival (30 days) studies performed with NMC Per403 and 11060 xenografts were initiated as above. For these studies, (+)-JQ1 was administered at a dose of 50 mg kg-1 by daily intraperitoneal injection. The average size of tumors in the (+)-JQ1 treatment group (n = 10) and vehicle group (n = 10) were similar at the start of treatment. Animals were followed daily for clinical symptoms. Tumor measurements were assessed by caliper measurements, and volume calculated using the formula Vol = 0.5xLxW2. Statistical significance of tumor volumes was calculated by two-sided Students t-test. Comparative survival analysis was performed using the Log-rank (Mantel-Cox) Test, and data were presented as a Kaplain-Meier plot annotated with a measure of statistical significance (pvalue). All animal studies were approved by the IACUC of the DFCI. \n\\nPharmacokinetic Studies in Mice.[1] Male CD1 mice (24 – 29 gm) were treated with a single dose of (+)-JQ1 at 5 mg kg-1 for intravenous tail vein injection studies and 10 mg kg-1 for oral gavage studies. Approximately 150 μl of blood were taken from animals by retro-orbital puncture under anesthesia with Isoflurane into EDTA tubes at pre-specified time intervals: 0.033, 0.083, 0.25, 0.5, 1, 2, 4, 5, 8 and 24 hours. Three animals were analyzed per time point. Blood samples were put on ice and centrifuged to obtain plasma samples (2000 x.g, 5 min under 4 °C) within 15 minutes post-sampling. Plasma samples were stored at approximately -70 °C until analysis was performed. Mice were provided free access to food and water throughout the study. Compound was formulated for intravenous injection in 10 % DMSO and 10 % HP-β-CD. Pharmcokinetic studies and pharmacologic assay development was performed at ChemPartner (Shanghai, CHINA). Data were analyzed by J.E.B. using Microsoft Excel and GraphPad Prism 5.02. \n\\n In vivo formulations used (reported): \n1. MOLM-13 AML xenograft model: Female nude mice (6–8 weeks old, 18–22 g) were subcutaneously injected with 5×10⁶ MOLM-13 cells (suspended in 0.2 mL PBS:Matrigel = 1:1) into the right flank. When tumors reached ~100 mm³, mice were randomized into 2 groups (n=6/group): \n - Vehicle group: 0.2 mL of 5% DMSO + 20% Cremophor EL + 75% saline, oral gavage, once daily for 21 days; \n - (+)-JQ1 group: 50 mg/kg (+)-JQ1 (dissolved in the above vehicle to 250 mg/mL), 0.2 mL oral gavage, once daily for 21 days. \n Tumor volume (length × width² / 2) and body weight were measured every 3 days. On day 22, mice were euthanized, and tumors were collected for mRNA and protein analysis [1] \n2. Male contraception mouse model: Male C57BL/6 mice (8 weeks old, 22–25 g) were randomized into 2 groups (n=8/group): \n - Vehicle group: 0.1 mL of 10% DMSO + 90% saline, intraperitoneal injection, once daily for 28 days; \n - (+)-JQ1 group: 25 mg/kg (+)-JQ1 (dissolved in the above vehicle to 250 mg/mL), 0.1 mL intraperitoneal injection, once daily for 28 days. \n After treatment, epididymides were collected to count sperm and measure motility. Testes were fixed for HE staining. Four weeks after drug withdrawal, sperm parameters were re-evaluated [3] |
| ADME/Pharmacokinetics |
The bromine-domain inhibitor (+)-JQ1 is a highly validated chemical probe; however, its in vivo pharmacokinetic properties are poor. The significant inter-individual variability in this small study may cast doubt on the significance of the average variability obtained from the pharmacokinetic analysis. Given that our primary objective was to assess the relative effect of deuteration on (+)-JQ1 clearance, rather than to determine the absolute pharmacokinetic parameters of (+)-JQ1 and (+)-JQ1-D, we examined the isotope ratios of (+)-JQ1 and its major metabolite M19 to look for evidence of deuteration. In both male and female mice, the (+)-JQ1/(+)-JQ1-D ratio consistently decreased over the time course examined (Figure 7), indicating that (+)-JQ1 was cleared faster than (+)-JQ1-D. [https://pmc.ncbi.nlm.nih.gov/articles/PMC10788937/]
Plasma pharmacokinetic studies were performed on non-tumor mice that received intraperitoneal injection of 30 mg/kg JQ1. All JQ1 concentrations collected at 6 hours post-administration were below the limit of quantitation (LOQ). A linear one-compartment model was used to fit the total plasma concentration-time data of JQ1 well, assuming that intraperitoneal absorption was immediate and complete (Figure 6). The estimated model parameters are shown in Table 7. The finite sampling model determined the following effective time points for plasma sampling during the brain microdialysis study: 15 minutes, 1 hour, and 6 hours post-administration. [https://pmc.ncbi.nlm.nih.gov/articles/PMC8384680/] 1. Oral bioavailability: Male SD rats (250–300 g, n=3 at each time point) were administered (+)-JQ1 by gavage (50 mg/kg) or intravenous injection (10 mg/kg). The oral bioavailability was calculated to be ~35% (based on AUC₀₋₂₄ₕ: oral ≈15 μM·h; intravenous ≈43 μM·h)[1] 2. Plasma pharmacokinetics: After oral administration (50 mg/kg) to rats, the key parameters were: Cₘₐₓ ≈ 2.8 μM (Tₘₐₓ = 1.5 h), terminal half-life (t₁/₂) ≈ 3.2 h, and clearance (CL) ≈ 18 mL/kg/min. After intravenous injection (10 mg/kg): Cₘₐₓ ≈ 12 μM, t₁/₂ ≈ 2.8 h, CL ≈ 15 mL/kg/min [1] 3. Tissue distribution: 1.5 h after oral administration of (+)-JQ1 (50 mg/kg) to rats, the concentrations detected by LC-MS/MS were: liver ≈ 4.5 μM, kidney ≈ 3.2 μM, MOLM-13 xenograft ≈ 3.8 μM, brain ≈ 0.3 μM (low blood-brain barrier penetration) [1] |
| Toxicity/Toxicokinetics |
1. Subchronic toxicity in rats: The weight change in rats orally administered (+)-JQ1 (50 mg/kg, 21 days) was less than that in the control group (<5%). Serum ALT/AST was approximately 1.1 times that of the control group (within the normal range), and creatinine levels were normal. Peripheral blood leukocyte count was approximately 0.9 times that of the control group (no significant difference) [1]
2. Testicular safety in mice: The seminiferous tubules of mice intraperitoneally injected with (+)-JQ1 (25 mg/kg, 28 days) were intact (HE staining), without inflammation or necrosis. Serum testosterone levels were approximately 0.9 times that of the carrier (no significant difference) [3] 3. Plasma protein binding rate: The plasma protein binding rate of (+)-JQ1 (1 μM) in human plasma was approximately 85% (measured using an ultrafiltration membrane with a molecular weight cutoff of 30 kDa and LC-MS/MS) [1] |
| References | |
| Additional Infomation |
JQ1 belongs to the thienotriazole diazapine class of compounds and is the tert-butyl ester of [(6S)-4-(4-chlorophenyl)-2,3,9-trimethyl-6H-thieno[3,2-f][1,2,4]triazolo[4,3-a][1,4]diazaphen-6-yl]acetic acid. It is a bromine domain protein 4 inhibitor with anticancer and cardioprotective effects. JQ1 can function as a bromine domain protein 4 inhibitor, cardioprotective agent, antitumor agent, anti-inflammatory agent, angiogenesis inhibitor, apoptosis inducer, and ferroptosis inducer. It is a thienotriazole diazapine, organochlorine compound, carboxylic acid ester, and tert-butyl ester. Epigenetic proteins are highly sought-after targets in ligand discovery. To date, successful studies have been limited to chromatin-modifying enzymes, the so-called epigenetic “writers” and “erectors.” No effective histone-binding module inhibitors have been reported. This article reports a cell-permeable small molecule (JQ1) that competitively binds to an acetyllysine recognition motif (bromodomain). Co-crystal structure analysis with BRD4, a member of the bromodomain and terminal extra-terminal domain (BET) family, revealed that JQ1 has excellent shape complementarity with the acetyllysine binding cavity, thus explaining its high efficiency and specificity for a specific human bromodomain subset. Recurrent translocation of BRD4 has been observed in a genetically determined, incurable subtype of human squamous cell carcinoma. Competitive binding of JQ1 can displace the BRD4 fusion oncoprotein from chromatin, thereby inducing squamous differentiation and producing specific antiproliferative effects in BRD4-dependent cell lines and patient-derived xenograft models. These data provide proof of concept for protein-protein interactions targeting epigenetic “readers” and provide a versatile chemical scaffold for the broader development of chemical probes for the bromodomain family. [1] Autophagy is a membrane transport process that directs the degradation of cytoplasmic material by lysosomes. The process promotes cell integrity, and although the core mechanism of autophagy has been elucidated, the mechanisms that promote and maintain autophagy are not fully understood. This paper reports that the epigenetic reader BRD4 and the methyltransferase G9a inhibit a transcriptional program that is independent of TFEB/TFE3/MITF, which promotes autophagy and lysosomal biosynthesis. We found that BRD4 knockdown can induce autophagy in vitro and in vivo under certain (but not all) conditions. Under starvation conditions, a signaling cascade involving AMPK and histone deacetylase SIRT1 dissociates chromatin-bound BRD4, thereby activating autophagy genes and promoting cell survival. Importantly, this process is independent of the growth-promoting properties of BRD4 and is antagonistic to it, and is strongly inhibited by the NUT midline oncology driver BRD4-NUT. Thus, these findings reveal a unique, selective mechanism of autophagy regulation. [2] The pharmacological treatment of male contraception has been a long-standing challenge in the medical field. To achieve this goal, we investigated the effects of a selective small molecule inhibitor (JQ1) on the bromodomain and end-external (BET) subfamily of epigenetic reading proteins. This paper reports a potent inhibitor of the testis-specific member BRDT, which plays a crucial role in chromatin remodeling during spermatogenesis. Biochemical and crystallographic studies confirmed that JQ1 occupies the acetyl-lysine binding pocket of BRDT, preventing recognition by acetylated histone H4. Treatment of mice with JQ1 reduced seminiferous tubule area, testis size, and sperm count and motility without affecting hormone levels. Although JQ1-treated male mice mated normally, the inhibitory effect of JQ1 at the spermatocyte and round spermatocyte stages resulted in complete and reversible contraceptive efficacy. These data suggest that JQ1 is a novel contraceptive capable of crossing the blood-testis barrier and inhibiting bromodomain activity during spermatogenesis, providing a lead compound for targeted contraception in male germ cells. [3] BET inhibition mechanism: (+)-JQ1 competitively binds to the acetyl-lysine binding pocket of the BET bromine domain (e.g., BRD4), preventing BET proteins from binding to chromatin. This blocks the transcription of BET-dependent oncogenes (e.g., MYC), which drive the proliferation of various cancers [1].
2. Autophagy activation mechanism: BRD4 negatively regulates autophagy by binding to the promoter of autophagy-related genes (e.g., ATG5) and inhibiting their transcription. (+)-JQ1 inhibits BRD4, unblocks this inhibition and activates autophagy, thereby enhancing the sensitivity of cancer cells to nutrient deprivation [2]. 3. Male contraceptive potential: BRDT is essential for spermatogenesis (expressed only in testicular germ cells during meiosis). (+)-JQ1 inhibits BRD4, blocking spermatogonia from differentiating into spermatocytes, resulting in reversible oligoasthenospermia. It avoids the hormonal side effects (unlike traditional contraceptives), making it a candidate for a non-hormonal male contraceptive method [3]. |
| Molecular Formula |
C23H25CLN4O2S
|
|
|---|---|---|
| Molecular Weight |
456.99
|
|
| Exact Mass |
456.138
|
|
| Elemental Analysis |
C, 60.45; H, 5.51; Cl, 7.76; N, 12.26; O, 7.00; S, 7.02
|
|
| CAS # |
1268524-70-4
|
|
| Related CAS # |
(R)-(-)-JQ1 Enantiomer;1268524-71-5;JQ-1 (carboxylic acid);202592-23-2
|
|
| PubChem CID |
46907787
|
|
| Appearance |
White to yellow solid
|
|
| Density |
1.3±0.1 g/cm3
|
|
| Boiling Point |
610.4±65.0 °C at 760 mmHg
|
|
| Flash Point |
322.9±34.3 °C
|
|
| Vapour Pressure |
0.0±1.7 mmHg at 25°C
|
|
| Index of Refraction |
1.657
|
|
| LogP |
4.49
|
|
| Hydrogen Bond Donor Count |
0
|
|
| Hydrogen Bond Acceptor Count |
6
|
|
| Rotatable Bond Count |
5
|
|
| Heavy Atom Count |
31
|
|
| Complexity |
706
|
|
| Defined Atom Stereocenter Count |
1
|
|
| SMILES |
ClC1C([H])=C([H])C(=C([H])C=1[H])C1C2C(C([H])([H])[H])=C(C([H])([H])[H])SC=2N2C(C([H])([H])[H])=NN=C2[C@]([H])(C([H])([H])C(=O)OC(C([H])([H])[H])(C([H])([H])[H])C([H])([H])[H])N=1
|
|
| InChi Key |
DNVXATUJJDPFDM-KRWDZBQOSA-N
|
|
| InChi Code |
InChI=1S/C23H25ClN4O2S/c1-12-13(2)31-22-19(12)20(15-7-9-16(24)10-8-15)25-17(11-18(29)30-23(4,5)6)21-27-26-14(3)28(21)22/h7-10,17H,11H2,1-6H3/t17-/m0/s1
|
|
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code |
2934.99.9001
|
|
| Storage |
Powder -20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
| Solubility (In Vitro) |
|
|||
|---|---|---|---|---|
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.47 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution.
For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.47 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. View More
Solubility in Formulation 3: ≥ 2.5 mg/mL (5.47 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. Solubility in Formulation 4: 2% DMSO+30% PEG 300+5% Tween 80+ddH2O:5mg/mL |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1882 mL | 10.9412 mL | 21.8823 mL | |
| 5 mM | 0.4376 mL | 2.1882 mL | 4.3765 mL | |
| 10 mM | 0.2188 mL | 1.0941 mL | 2.1882 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles.
Calculation results
Working concentration: mg/mL;
Method for preparing DMSO stock solution: mg drug pre-dissolved in μL DMSO (stock solution concentration mg/mL). Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug.
Method for preparing in vivo formulation::Take μL DMSO stock solution, next add μL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O,mix and clarify.
(1) Please be sure that the solution is clear before the addition of next solvent. Dissolution methods like vortex, ultrasound or warming and heat may be used to aid dissolving.
(2) Be sure to add the solvent(s) in order.
Leukemia and lymphoma cell lines are broadly sensitive to BET-bromodomain inhibition.Proc Natl Acad Sci U S A.2011 Oct 4;108(40):16669-74. |
Gene expression profiling of LP-1 and Raji cells treated with active or inactive BET inhibitors.Proc Natl Acad Sci U S A.2011 Oct 4;108(40):16669-74. |
Small molecule BET-bromodomain inhibition suppressesMYCtranscription.Proc Natl Acad Sci U S A.2011 Oct 4;108(40):16669-74. |
MYC reconstitution significantly protects cells from BET-mediated effects.Proc Natl Acad Sci U S A.2011 Oct 4;108(40):16669-74. |
BET-bromodomain inhibition decreases tumor load in vivo.Proc Natl Acad Sci U S A.2011 Oct 4;108(40):16669-74. |
Integrated genomic rationale for BET bromodomains as therapeutic targets in MM.Cell.2011 Sep 16;146(6):904-17. |
Inhibition of Myc-dependent transcription by theJQ1BET bromodomain inhibitor.Cell.2011 Sep 16;146(6):904-17. |
BET inhibition suppressesMYCtranscription in MM.Cell.2011 Sep 16;146(6):904-17. |
Regulation ofMYCtranscription by BET bromodomains.Cell.2011 Sep 16;146(6):904-17. |
Anti-myeloma activity ofJQ1in vitro.Cell.2011 Sep 16;146(6):904-17. |
JQ1induces cell cycle arrest and cellular senescence in MM cells.Cell.2011 Sep 16;146(6):904-17. |
Translational implications of BET bromodomain inhibition in MM.Cell.2011 Sep 16;146(6):904-17. |
|
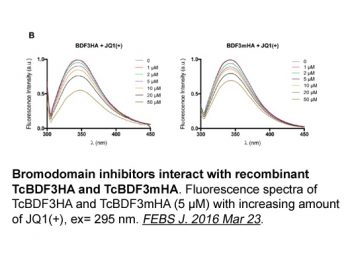 |
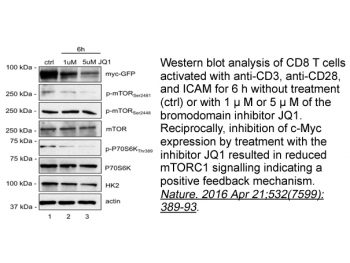 |